
As seawater begins to freeze, crystals start forming from water molecules. In this process, impurities such as salts and minerals get expelled from the ice, creating a porous structure. Within these porous cavities, trapped seawater can become highly saturated in seasalt, forming a liquid known as brine. [1]
This process has important physical consequences, as liquids with a higher salt concentration have a significantly lower freezing point. This is because the dissolved minerals hinder the formation of ice crystals and permit the brine to enter a supercooled state, remaining liquid even well below the usual freezing temperature of seawater. Under the pack ice, the seawater temperature remains relatively stable at about −2 °C, whereas the brine trapped in the ice can cool down to temperatures as low as −20 °C.
At the same time, its high salt content makes the brine significantly denser than the surrounding water. Thus, as the brine is heavier than regular seawater, it can start to leak out of the ice and form a downward-flowing jet. As the brine creeps towards the sea bed, it comes into contact with the relatively warmer seawater. The extreme temperature difference causes the surrounding water to freeze instantly, forming a hollow tube of ice around the descending flow. In a sense, you could thus consider a brinicle to be an inverted icicle. While ordinary icicles, or ice-stalactites, form when cold air freezes downwards-flowing water in layers, brinicles grow as extremely cold descending liquid freezes the surrounding seawater. This is why scientists actually prefer the term brinicle over “sea-ice stalactite.”[1]
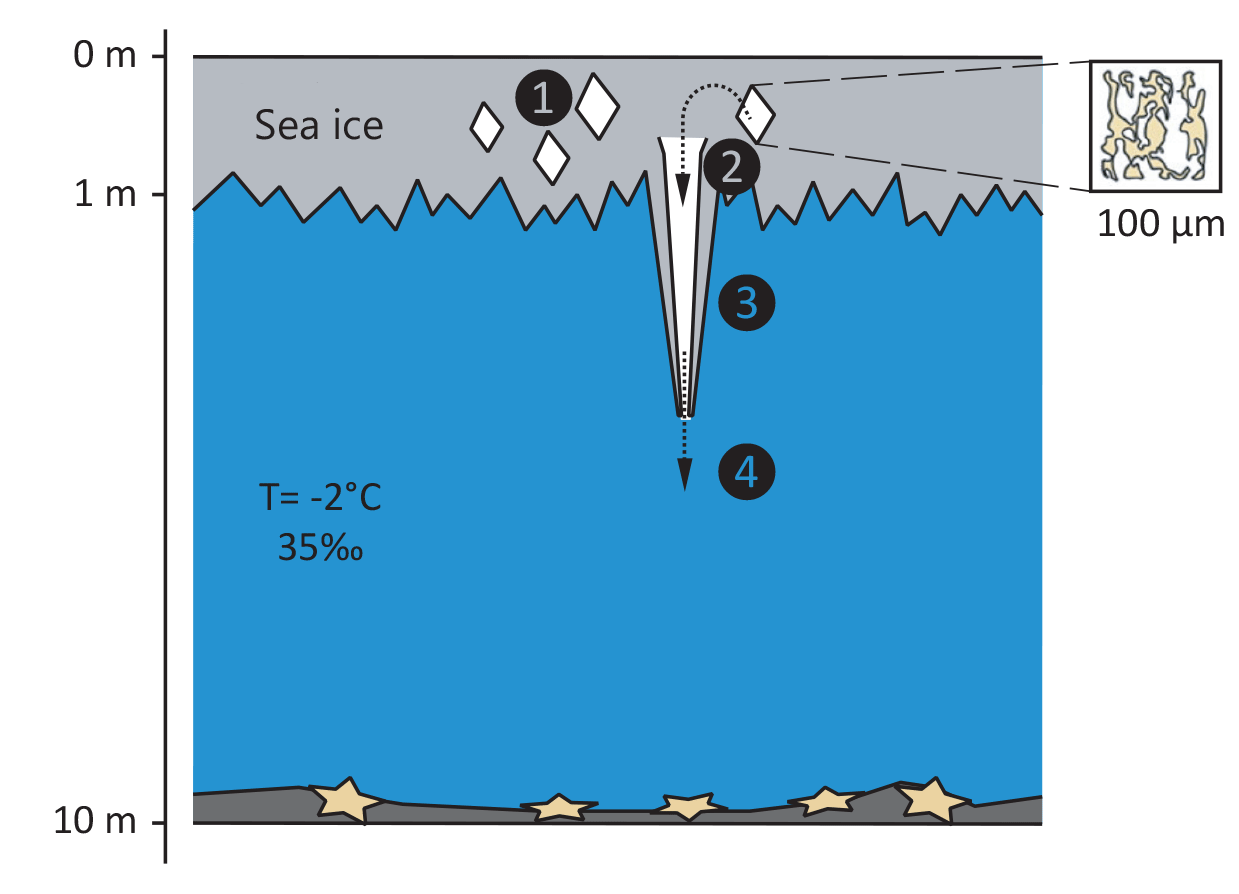
At the beginning of their formation, brinicles have thin walls and are fragile. However, under the right circumstances; i.e., minimal ocean current and suitable temperature, salinity, and depth conditions; a steady flow of supercooled brine can thicken the ice walls and stabilise the structure. The tube of ice surrounding the brine moreover acts as insulation which reduces heat exchange with the warmer surrounding seawater. This causes the brine to remain in its supercooled state and allows the brinicle to continue growing downward as long as the flow persists.
In this way, brinicles can sometimes grow all the way down to the sea bed, leading to a rather dramatic phenomenon known as the “finger of death.” As the brinicle extends, it resembles a long icy finger reaching downward. When it finally touches the ground, the brine continues to leak from the column and flows outwards along the ocean floor. Because this brine is so much colder, it immediately freezes the surrounding seawater upon contact, thus creating a spreading sheet of ice. Unfortunately, unsuspecting marine animals like sea urchins and starfish are adapted to relatively stable temperatures at the seafloor and can thus become trapped in this rapidly forming ice-web and freeze.
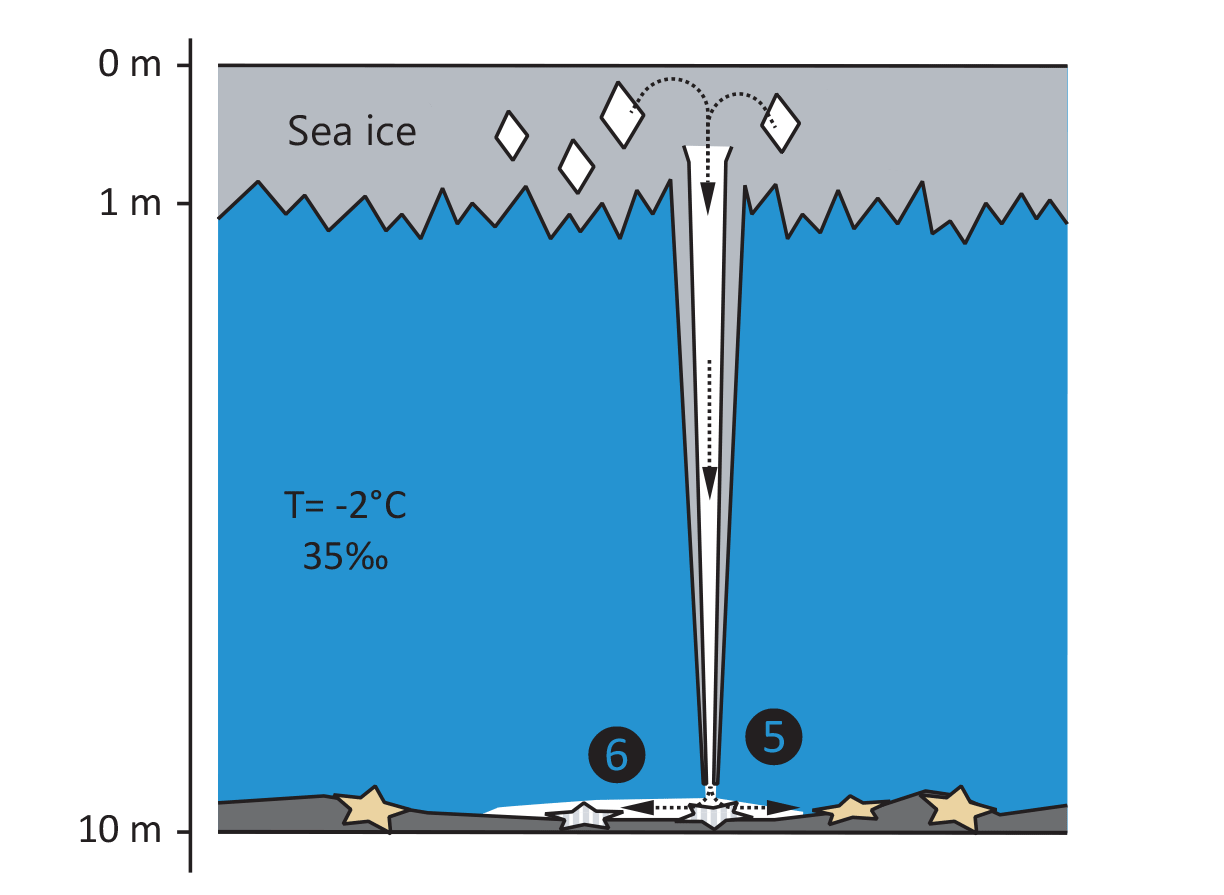
The spectacular phenomenon of the “finger of death” was captured in the BBC documentary Frozen Planet in 2011. You can skip ahead to 1m45s in the video below to see the brinicle grow and perform its “touch of death” in real time. Remarkably, although brinicles have reportedly been studied since the 1970s [2], this 2011 documentary was the first instance in which divers achieved to fully catch the phenomenon on camera [3].
References
[1] Cartwright, J.H., Escribano, B., González, D.L., Sainz-Díaz, C.I. and Tuval, I., 2013. Brinicles as a case of inverse chemical gardens. Langmuir, 29(25), pp.7655-7660.
[2] Testón-Martínez, S., Barge, L.M., Eichler, J., Sainz-Díaz, C.I. and Cartwright, J.H., 2024. Experimental modelling of the growth of tubular ice brinicles from brine flows under sea ice. The Cryosphere, 18(5), pp.2195-2205.
[3] BBC: Finger of death. BBC Frozen Planet (Winter), https://www.bbc.co.uk/programmes/p00mq92j, 2011