
In my previous article, Creating new colours with lasers, I explained how different wavelengths of the electromagnetic spectrum correspond to certain colours we can see. I also described how the human eye detects these colours thanks to specialised cone cells in the retina; these respond to red, green, and blue wavelengths. Our brain then combines these signals to produce the full colour-range we experience. This is why most people can perceive not only so-called primary colours, like red or blue, but also mixtures such as magenta or cyan. However, this raises a deeper question: Beyond our perception, why do objects and substances have specific colours at all? What determines whether something appears blue, red, or green in the first place?
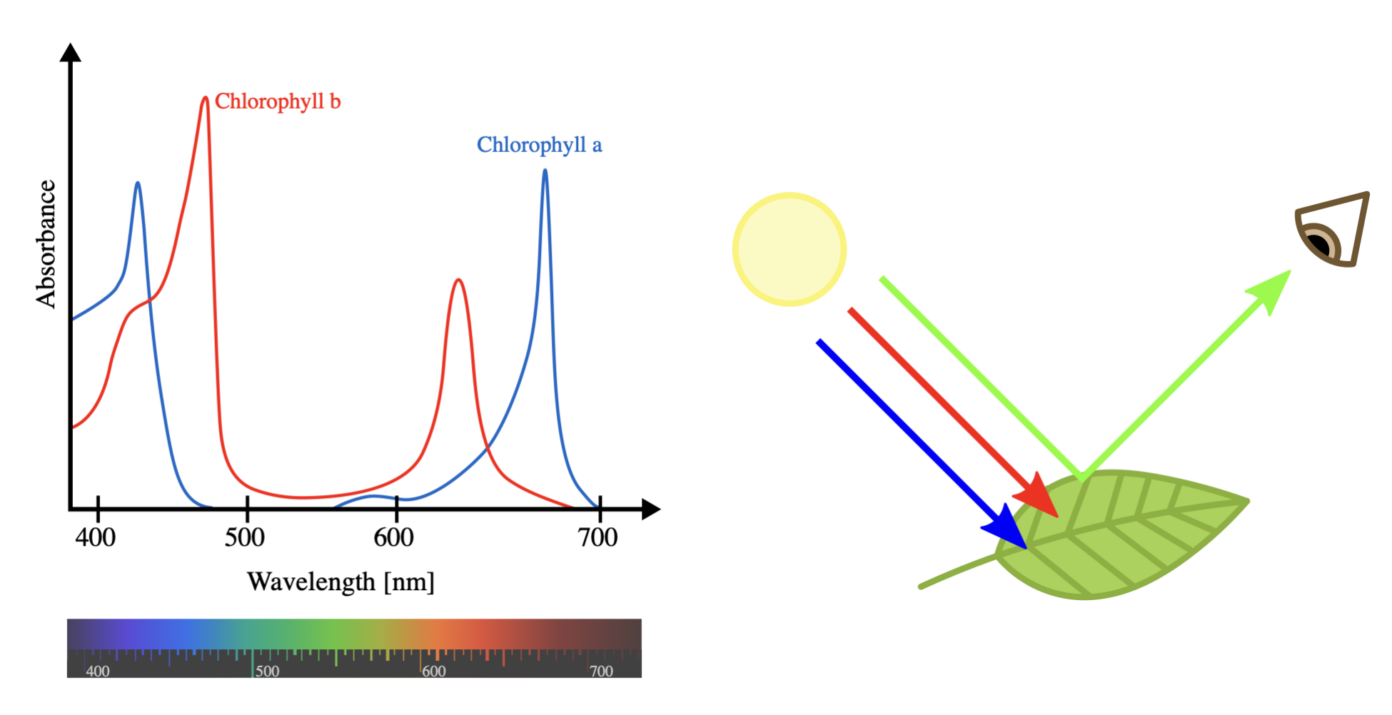
In this older article, Jans Henke explained several mechanisms by which colour can arise in nature. The most straightforward one is based on absorption and reflection. You might have heard that “white” light, such as sunlight, contains a mixture of all the colours of the rainbow. When this light hits an object, some wavelengths are absorbed while others are reflected, depending on the material and, for instance, any pigments it may contain. This explains why plants appear green: different types of chlorophyll pigments, which are essential for photosynthesis in plants, absorb much of the red and blue light, while reflecting green light back to our eyes.
However, pigments are not the only way colour can appear. In particular, true blue pigments are very rare in nature, yet many animals, such as certain butterflies and birds, display striking blue colours due to structural colouration. As explained in the same QU article, microscopic structures on wings or feathers can scatter and interfere with light in such a way that only certain wavelengths, often blue, are strongly reflected.
A different effect, called Rayleigh scattering, explains why the sky appears blue. As Jorrit Kruthoff describes here, shorter wavelengths of light, like blue, are scattered more strongly than longer wavelengths when sunlight passes through air and hits small suspended particles. This is why blue light is spread across the sky. A similar scattering effect is even responsible for the appearance of blue eyes, which actually do not contain any blue pigment at all.
So, blue colour can arise in several different ways: through pigments, through interference with microscopic structures, or through scattering in a medium. This raises an interesting question. Large bodies of water, such as the sea, often appear deep blue, yet a glass of water looks completely clear. If water has no obvious pigment and no visible structure, what is it that gives the ocean its colour?
Based on what we know about how blue colour can arise, one might guess that the ocean appears blue because blue light is scattered from its surface, while other colours are absorbed. There is indeed some scattering at the surface, similar to what happens in the atmosphere. However, this idea alone is inconsistent with what you can observe when you open your eyes underwater in a pool or the sea. You may have noticed that everything takes on a more blue or green tint, especially as you dive deeper. However, this could not possibly be the case if all the blue light were simply scattered away at the surface. So what is really going on?
The actual answer is much deeper and based on the way water interacts with incident light. Water is a transparent but dense medium, thus light is gradually absorbed and scattered as it travels through. Crucially, these phenomena do not happen equally for all colours. You can think of water as a filter: the further light travels through it, the more certain colours from the spectrum are “removed” (i.e. filtered out).
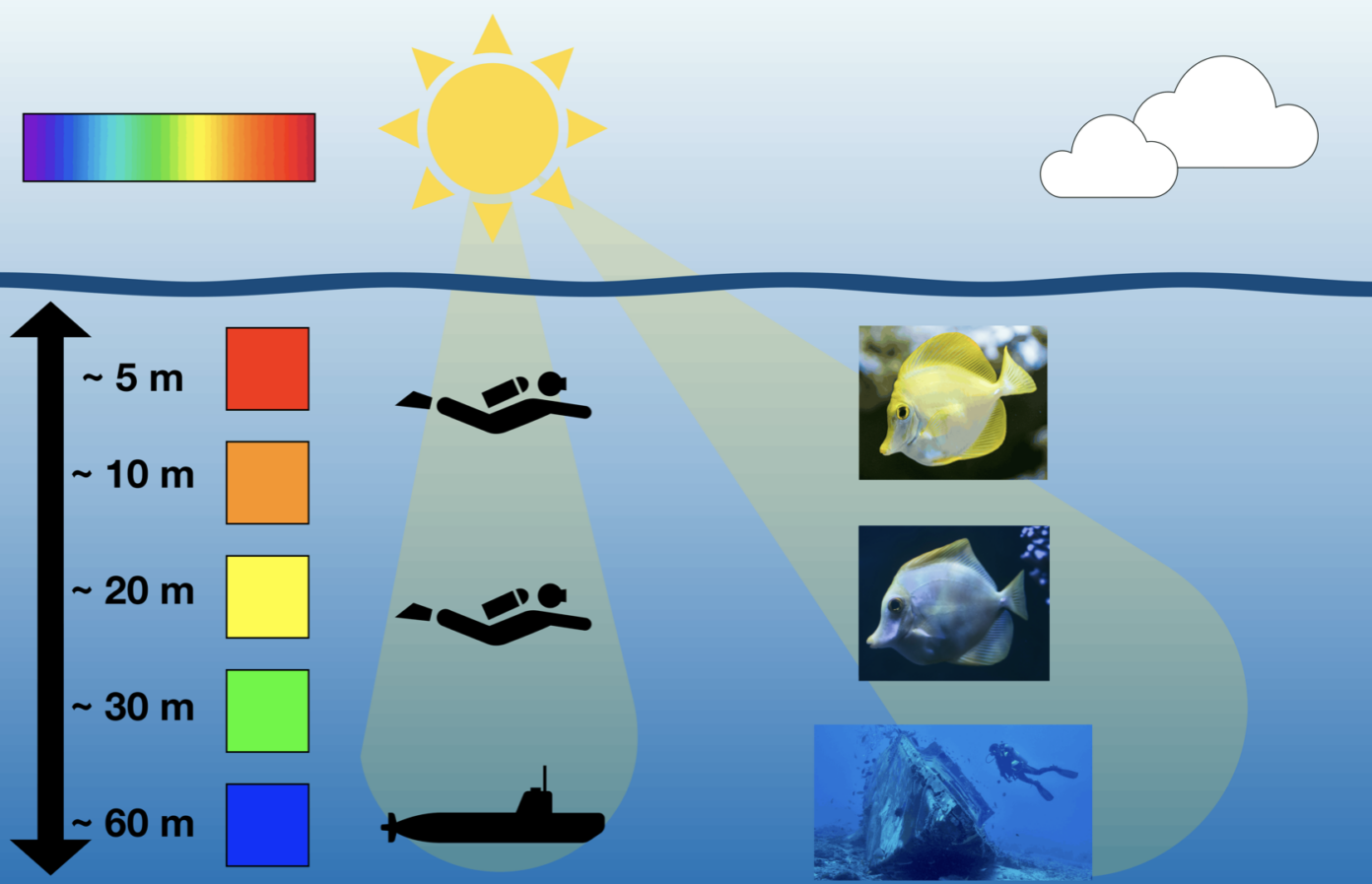
Due to this filtering effect, different wavelengths can penetrate to different depths. Red and orange light are absorbed most strongly and disappear first, while shorter wavelengths, such as blue, can travel much further. As a result, the remaining light becomes increasingly blue the deeper you go or the further you look through the water. More specifically, red light is largely absorbed only a few metres below the surface (around 5 m), followed by orange (around 10 m), yellow (around 20 m), and green (around 30 m), while blue light can reach depths of up to about 60 m. [1]
This is why you notice colour changes when swimming, snorkelling, or diving. Near the surface, colours appear much as they do in air. But as you descend, or even when you simply look across a distance underwater, colours begin to fade and shift towards duller, brownish tones. In a coral reef, for example, fish and corals may display vivid colours for signalling, camouflage, or attracting mates, yet these colours quickly lose their brightness with depth.[2] As you go deeper, entire colours vanish. For instance, if you were to cut yourself at a depth of around 20 m, your blood would appear greenish rather than bright red.
In the upper 200 metres of the ocean there is enough sunlight present for organisms like plants, algae, or phytoplankton to do photosynthesis, which is why this is also known as “photic” or sunlight zone. Diving deeper, one enters the “dysphotic” or twilight zone where only a small fraction of sunlight is left. Ultraviolet and infrared radiation does not reach these depths and the remaining blue-ish visible light is so dim that photosynthesis becomes impossible. Eventually, at depths below 1000 metres, all sunlight is absorbed, and the ocean becomes completely dark. In this “aphotic” zone, the only remaining light source is bioluminescence, i.e., light emitted by biochemical processes in living organisms like the angler-fish. [3]
I should note one additional effect that we have largely neglected so far. In reality, the colour of natural bodies of water is also influenced by small particles suspended in it, like algae, sand, or mud. [4] These particles scatter and absorb light in different ways, often giving the water a green or brown appearance. This is why sea-water in coastal regions or sediment-rich rivers, such as the Amazon, can look green or milky brown, rather than blue.
So, to answer our initial question about why the ocean looks blue when viewed from above: The key idea is that water gradually filters out the red and yellow parts of sunlight as it travels through it, while blue light remains and is scattered back towards our eyes. Combined with a small contribution from the reflection of the sky, this gives the ocean its characteristic deep blue colour.
After learning about the electromagnetic spectrum and how visible light interacts with water, you might be tempted to think that higher-energy light always penetrates further than lower-energy light. Within the visible range, this seems to hold: blue light, which has a shorter wavelength and higher frequency, travels deeper than red light. However, this pattern does not apply universally. The way electromagnetic radiation is absorbed by water is much more complex, as shown in the absorption curve below.
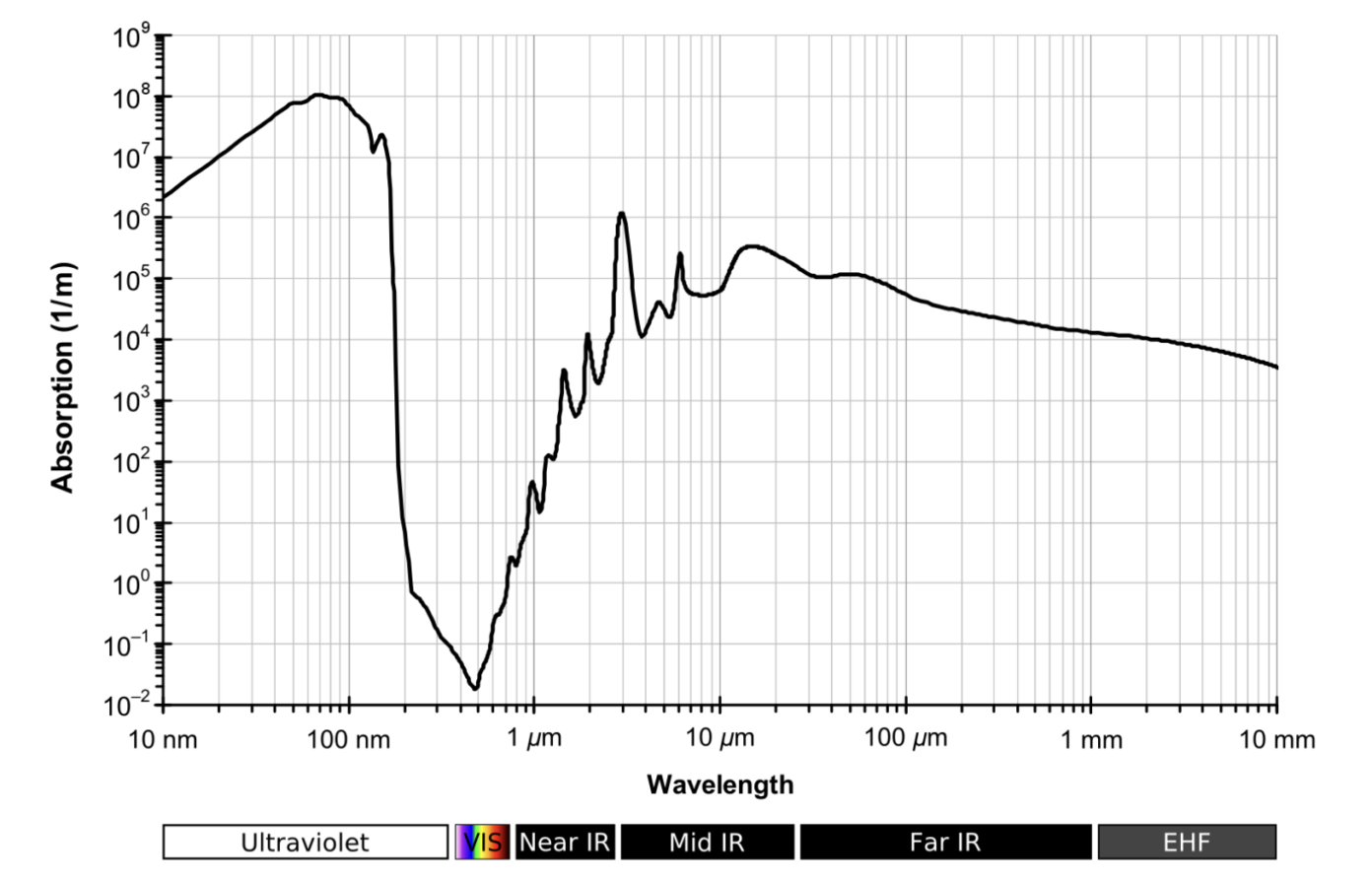
From the absorption curve, we can see that liquid water has a rather special property: it absorbs visible light only weakly compared to many other parts of the spectrum. This is what allows light to penetrate the ocean at all, whereas ultraviolet (UV) and infrared (IR) light get absorbed very quickly, thus preventing them from reaching deeper layers of water.
To understand why this happens, we need to look at how light interacts with molecules. Recall that according to quantum mechanics, light comes in particles – photons – and that the frequency (and therefore the wavelength) of the light determines the energy of the photon. When a photon of light of a given wavelength encounters a water molecule, it can transfer its energy to that molecule. If the energy (i.e., frequency) of the photon matches one of the molecule’s natural modes of motion, such as vibrations or rotations, the molecule can absorb the incoming light. You can think of this a bit like pushing a child on a swing: if you push at just the right rhythm (i.e. frequency), the energy gets “absorbed”, making the child swing higher and higher.
In the liquid state, water molecules can move, rotate, and vibrate. If a molecule is hit by a photon from the IR part of the spectrum, it can absorb a quantum of rotational or vibrational energy. Moreover, photons from the UV part of the electromagnetic spectrum can hit the water molecules in precisely such a way that electrons can jump to a higher energy level. Thus, infrared and ultraviolet photons match certain frequencies that are characteristic of molecules in liquid water, which makes it strongly absorbing in these parts of the electromagnetic spectrum. However, visible light happens to sit exactly in an “energy gap” region between these two regimes. Therefore, it is too energetic to excite the vibrational or rotational modes but not quite energetic enough to trigger the electronic transitions caused by UV light. This is the reason why liquid water is relatively transparent to visible light, with blue light being the one that interacts the least with the water molecules. Thus, blue is the colour that remains the most visible under water, especially after the light has travelled over longer distances and “lost” its other components due to different absorption processes.
References
[1] https://thescubadirectory.com/blog-post/scuba-diving/science-of-light-color-scuba-diving
[2] https://marinedynamics.org/academy/2018/11/05/looking-into-the-underwater-world-how-colour-is-perceived-beneath-the-waves; Article by Valerio Tettamanti (Nov 5 2018); “Looking into the Underwater World – How Colour is Perceived Beneath the Waves”
[4] https://www.whoi.edu/oceanus/feature/shedding-light-on-light-in-the-ocean/